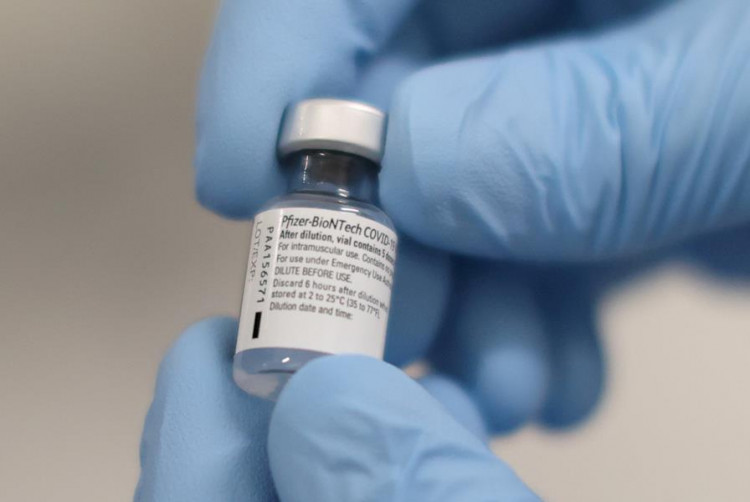
A U.S. Food and Drug Administration panel has recommended Pfizer, Inc. and BioNTech SE's vaccine be given the go for emergency use, The Associated Press reported Friday.
The 17-4 vote might result in the administration authorizing the treatment within a matter of days - which would then set the stage for the vaccine's distribution to front-line health workers, pharmacies and nursing home residents.
The recommendation came as COVID infections climb ever higher across the U.S. - with single-day deaths reaching more than 3,100 Wednesday.
The priority for distribution is to get around 3 million doses of the vaccine to cover the 64 jurisdictions in the U.S., including 50 states, Tribal Nations and multiple government territories.
Albert Bourla, chief executive officer of Pfizer, applauded the decision. "We're happy with the strong majority vote and if the administration issues an authorization, stand ready to bring this vaccine to people across the U.S.," CBS News quoted him saying.
The decision is "a huge milestone," Dr. Ofer Levy, a pediatric infectious disease expert, said. Levy is also director of the precision vaccine program at Boston Children's Hospital.
If the administration accords emergency approval, the U.S. will be the third country to authorize the use of the Pfizer/BioNTech treatment after Canada and the UK.
U.S. hospitals are nearing capacity at intensive care units. Almost 300,000 Americans have died. The disease has infected more than 15 million people in the U.S., data by Johns Hopkins University show.
Meanwhile, Dr. Robert Redfield said COVID had become the main cause of fatalities in the U.S. - exceeding heart disease. Redfield is chief of the U.S. Center for Disease Control and Prevention.