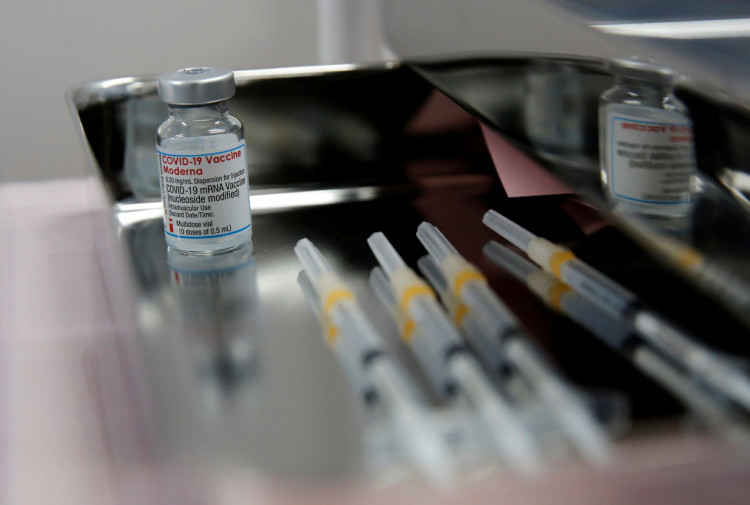
After months of waiting, a panel of experts convened by the U.S. Food and Drug Administration on Wednesday recommended Moderna's COVID-19 vaccine for the nation's youngest children.
The committee's recommendation is an essential step toward immunizing children under the age of five and as young as six months who have not yet been eligible for vaccinations.
Senior FDA scientist Peter Marks opened the meeting by saying that, despite studies showing that the majority of children are now infected with the coronavirus, the high rate of hospitalizations among infants, toddlers, and young children during last winter's Omicron wave highlighted the urgent need for vaccination.
According to the most recent official data, the U.S. has had 480 Covid-19 deaths in the 0-4 age group so far in the pandemic, considerably more than even a "bad flu season" like 2009-2010, when the H1N1 influenza strain caused 78 deaths in that age range, according to Marks.
Both vaccines use messenger RNA to send genetic code for the coronavirus spike protein to human cells, preparing the immune system for when it encounters the genuine virus. The technique is currently widely recognized as the most effective Covid immunization platform.
Pfizer wants the FDA to approve three doses of three micrograms for children aged six months to four years, whereas Moderna wants the FDA to approve two doses of a higher 25 micrograms for children aged six months to five years.
Both vaccines were tested on thousands of children in clinical trials. They were discovered to cause-related levels of mild side effects in older age groups and to elicit comparable levels of antibodies.
After two doses, four weeks apart, Moderna's vaccine should provide strong protection against severe disease, and the company is looking into adding a booster that would increase efficacy levels against mild disease.
However, when compared to Pfizer, Moderna's decision to use a higher dose is associated with higher levels of fever in response to the vaccine.
Pfizer's efficacy against infection was higher, with the company putting it at 80%, compared to Moderna's estimates of 51% for children aged six months to two years and 37% for those aged two to five years.
However, the Pfizer figure is based on a small number of cases and is thus considered preliminary. It also requires three doses to provide protection, with the third shot administered eight weeks after the second, which is administered three weeks after the first.
Although obesity, neurological disorders, and asthma are associated with an increased risk of severe disease in young children, severe outcomes are difficult to predict.
In addition, children can develop multisystem inflammatory syndrome in children (MIS-C), a rare but serious post-viral condition. Three to six percent of people can have COVID-19 symptoms for more than 12 weeks.